Description
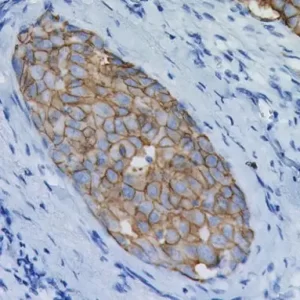
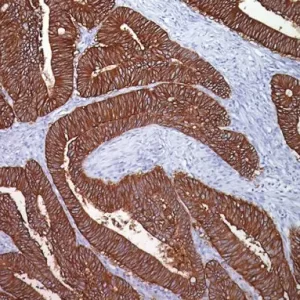
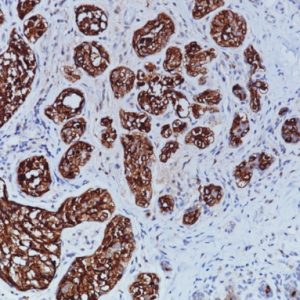
Uroplakin II [BC21] and Uroplakin III [BC17] are highly specific antibodies that may be useful in identifying tumors of urothelial origin. With the exception of bladder and ureter, staining was highly specific in various normal and neoplastic tissues in an in-house study. Both antibodies exhibited increased staining sensitivity when compared to Uroplakin III [AU1] in cases of urothelial carcinoma of the bladder. Uroplakin II + Uroplakin III antibody may be a specific and sensitive antibody cocktail for urothelial carcinoma and in discriminating bladder cancer from renal and prostate carcinomas. PATENT PENDING.
SPECIFICATIONS
Specifications
| INTENDED USE | IVD |
|---|---|
| FORMAT | Predilute, VALENT |
| SOURCE | Mouse Monoclonal |
| CLONE | BC21, BC17, Biocare Clone |
| ISOTYPE | IgG1 |
| ANTIGEN | Residues 36-50, Uroplakin III |
| LOCALIZATION | Cytoplasmic, Membrane |
| SPECIES REACTIVITY | Human |
DATA SHEET & SDS
REFERENCES
1. Wu XR, et al. Uroplakins in urothelial biology, function, and disease. Kidney Int.
2009 Jun; 75(11):1153-65.
2. Moll R, et al. Uroplakins, specific membrane proteins of urothelial umbrella cells, as
histological markers of metastatic transitional cell carcinomas. Am J Pathol. 1995 Nov;
147(5):1383-97.
3. Kaufmann O, Volmerig J, Dietel M. Uroplakin III is a highly specific and
moderately sensitive immunohistochemical marker for primary and metastatic
urothelial carcinomas. Am J Clin Pathol. 2000 May; 113(5):683-7.
4. Olsburgh J, et al. Uroplakin gene expression in normal human tissues and locally
advanced bladder cancer. J Pathol. 2003 Jan; 199(1):41-9.
5. Huang HY, et al. Persistent uroplakin expression in advanced urothelial carcinomas:
implications in urothelial tumor progression and clinical outcome. Hum Pathol. 2007
Nov; 38(11):1703-13.
6. Hoang LL, et al. A newly developed Uroplakin II antibody with increased sensitivity
in urothelial carcinoma of the bladder. Arch Pathol Lab Med. 2013; in press.
7. Center for Disease Control Manual. Guide: Safety Management, NO. CDC-22,
Atlanta, GA. April 30, 1976 “Decontamination of Laboratory Sink Drains to Remove
Azide Salts.”
8. Clinical and Laboratory Standards Institute (CLSI). Protection of Laboratory
Workers from Occupationally Acquired Infections; Approved Guideline-Fourth
Edition CLSI document M29-A4 Wayne, PA 2014.




Reviews
There are no reviews yet.