Description
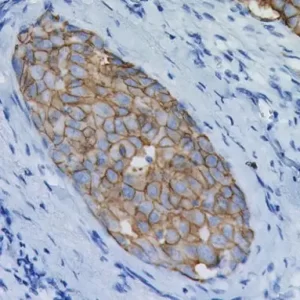
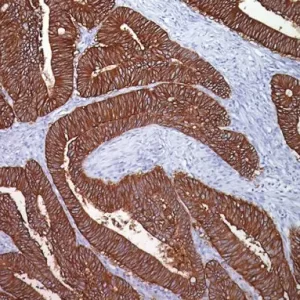
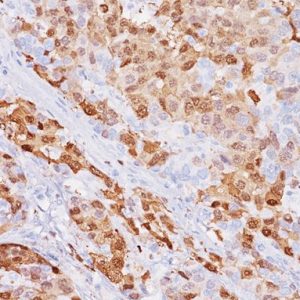
V-domain Ig-containing suppressor of T cell activation (VISTA), also known as PD-1H, is a member of the immunoglobulin (Ig) superfamily, whose extracellular domain bears homology to the B7 family ligand, particularly PDL1. VISTA is predominantly expressed on granulocytic, myeloid cells and T cells.15 VISTA expresses on T cells as a part of an inhibitory immune checkpoint that suppresses T cell activation, proliferation, and cytokine production.16 Recently, a number of research studies have reported the expression of VISTA in solid tumors such as melanoma, lung, pancreas and colorectal, these studies found that VISTA may act as an important biomarker in solid tumors.15,17
In both normal and malignant tissue, VISTA is expressed abundantly in vascular endothelial cells (noted as vasculature). Additionally, VISTA is typically expressed in quiescent naive T-Cells and monocytes and promotes quiescence in surrounding immune cells. Activated T-cells are reported to express less VISTA than quiescent cells.18 In our evaluation, tumor cells expressing VISTA were rare, however the tumor micro-environment showed a high prevalence of mixed-strength VISTA expression in vasculature, tumor infiltrating lymphocytes, and other tumors cells including but not limited to fibroblasts.
SPECIFICATIONS
Specifications
| INTENDED USE | IVD |
|---|---|
| FORMAT | Concentrate, Predilute |
| VOLUME | 0.1 ml, 1.0 ml, 6.0 ml |
| CLONE | BLR035F |
| SOURCE | Rabbit Monoclonal |
| SPECIES REACTIVITY | Human; others not tested |
| ISOTYPE | IgG |
DATASHEET & SDS
REFERENCES
1. Kiernan JA. Histological and Histochemical Methods: Theory and Practice. New York: Pergamon Press 1981.
2. Sheehan DC and Hrapchak BB. Theory and Practice of Histotechnology. St. Louis: C.V. Mosby Co. 1980.
3. Clinical Laboratory Improvement Amendments of 1988: Final Rule, 57 FR 7163, February 28, 1992.
4. Shi S-R, Cote RJ, Taylor CR. J Histotechnol. 1999 Sep;22(3):177-92.
5. Taylor CR, et al. Biotech Histochem. 1996 Jan;71(5):263-70.
6. Center for Disease Control Manual. Guide: Safety Management, NO. CDC-22, Atlanta, GA. April 30, 1976 “Decontamination of Laboratory Sink Drains to Remove Azide Salts.”
7. Clinical and Laboratory Standards Institute (CLSI). Protection of Laboratory Workers from Occupationally Acquired Infections; Approved Guideline-Fourth Edition CLSI document M29-A4 Wayne, PA 2014.
8. CLSI Quality Standards for Design and Implementation of Immunohistochemistry Assays; Approved Guideline-Second edition (I/LA28-A2) CLSI Wayne, PA USA (www.clsi.org). 2011
9. College of American Pathologists (CAP) Certification Program for Immunohistochemistry. Northfield IL. Http://www.cap.org (800) 323- 4040.
10. O’Leary TJ, Edmonds P, Floyd AD, Mesa-Tejada R, Robinowitz M, Takes PA, Taylor CR. Quality assurance for immunocytochemistry; Proposed guideline. MM4-P. National Committee for Clinical Laboratory Standards (NCCLS). Wayne, PA. 1997;1-46.
11. Koretzik K, Lemain ET, Brandt I, and Moller P. Metachromasia of 3- amino-9-ethylcarbazole (AEC) and its prevention in Immunoperoxidase techniques. Histochemistry 1987; 86:471-478.
12. Nadji M, Morales AR. Immunoperoxidase, part I: the techniques and its pitfalls. Lab Med 1983; 14:767.
13. Omata M, Liew CT, Ashcavai M, Peters RL. Nonimmunologic binding of horseradish peroxidase to hepatitis B surface antigen: a possible source of error in immunohistochemistry. AmJ Clin Path 1980; 73:626.
14. Herman GE and Elfont EA. The taming of immunohistochemistry: the new era of quality control. Biotech & Histochem 1991; 66:194.
15. He XL, Zhou Y, Lu HZ, Li QX, Wang Z. Prognostic value of VISTA in solid tumours: a systematic review and meta-analysis. Sci Rep. 2020;10(1):2662. Published 2020 Feb 14.
16. Hou Z, Pan Y, Fei Q, et al. Prognostic significance and therapeutic potential of the immune checkpoint VISTA in pancreatic cancer. J Cancer Res Clin Oncol. 2021;147(2):517-531.
17. Tagliamento M, Agostinetto E, Borea R, et al. VISTA: A Promising Target for Cancer Immunotherapy? Immunotargets Ther. 2021; 10:185-200. Published 2021 Jun 22.
18. Yuan, Long et al. VISTA: A Mediator of Quiescence and a Promising Target in Cancer Immunotherapy. Trends in immunology. 2021; 42(3): 209-227 DOI:10.1016/j.it.2020.12.008. Published 2021 Jan 23





Reviews
There are no reviews yet.