Description
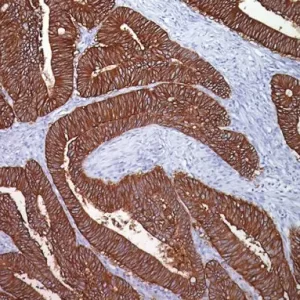
Prostate-specific acid phosphatase (PSAP) is an enzyme produced in prostate epithelial cells. PSAP expression levels proportionally increase with prostate cancer progression (1). PSAP IHC staining is often used in conjunction with prostate specific antigen (PSA) staining to help distinguish poorly differentiated carcinomas. For instance, PSAP is commonly used to help differentiate prostate adenocarcinoma and urothelial carcinoma which may appear microscopically similar; prostate adenocarcinoma often stains with PSA and/or PSAP, while urothelial carcinoma does not (2). PSAP has a significantly higher correlation with the morphological characteristics of prostate cancer and can provide a more accurate predictive prognosis than other markers currently available. Since PSAP detection is a proportional measure of prostate cancer progression, it can also be used as an immunotherapy target for treatment of prostate cancer (1). Due to its prostate specificity, PSAP may also be a useful marker for excluding metastases from a prostatic primary, particularly in male breast cancer (3).
Want to Learn More About PSAP Antibody? Download Our Complimentary White Paper!
SPECIFICATIONS
Specifications
| INTENDED USE | IVD |
|---|---|
| FORMAT | Concentrate, ONCORE Pro, Predilute, Q Series, UltraLine |
| VOLUME | 0.1 ml, 1.0 ml, 6.0 ml, 60 Tests, 7 ml |
| CLONE | rACPP/1338 |
| SOURCE | Mouse Monoclonal |
| SPECIES REACTIVITY | Human; others not tested |
| ISOTYPE | IgG1, Kappa |
| POSITIVE CONTROL | Normal prostate, prostate carcinoma |
DATASHEETS & SDS
REFERENCES
1. Kong HY, Byun J. Emerging roles of human prostatic acid phosphatase.
Biomol Ther (Seoul). 2013;21(1):10-20.
2. Genega EM, Hutchinson B, Reuter VE, Gaudin PB. Immunophenotype of
high-grade prostatic adenocarcinoma and urothelial carcinoma. Mod Pathol.
2000 Nov;13(11):1186-91.
3. Kidwai N, Gong Y, Sun X, et al. Expression of androgen receptor and
prostate-specific antigen in male breast carcinoma. Breast Cancer Res.
2004;6(1): R18-R23.
4. Center for Disease Control Manual. Guide: Safety Management, NO. CDC-
22, Atlanta, GA. April 30, 1976 “Decontamination of Laboratory Sink Drains
to Remove Azide Salts.”
5. Clinical and Laboratory Standards Institute (CLSI). Protection of
Laboratory Workers from Occupationally Acquired Infections; Approved
Guideline-Fourth Edition CLSI document M29-A4 Wayne, PA 2014.






Reviews
There are no reviews yet.