Description
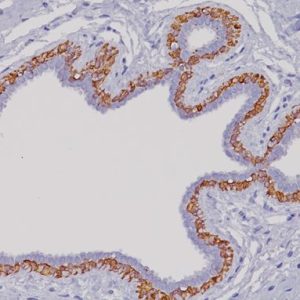
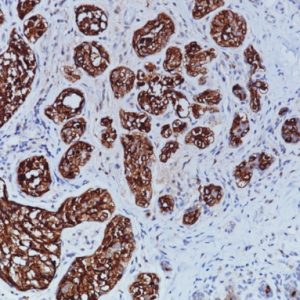
Pan Cytokeratin antibody Plus is a combination of [AE1/AE3] and Cytokeratin (CK) 8/18 [5D3] and can be used to detect most human epithelia. [AE1/AE3] recognizes acidic and basic subfamilies of cytokeratins, with molecular weights ranging from 40 to 67 kDa. CK8/18 [5D3] recognizes Cytokeratin 8 and 18 intermediate filament proteins. In normal tissues, [5D3] recognizes all simple and glandular epithelium. It has been observed that [AE1/AE3] has had problems marking certain tissues types and adenocarcinomas. The addition of CK 8/18 may remedy some of the limitations observed when staining with [AE1/AE3] alone.
SPECIFICATIONS
Specifications
| WEIGHT | N/A |
|---|---|
| DIMENSIONS | N/A |
| INTENDED USE | IVD |
| SPECIES REACTIVITY | Human, Mouse, Rat |
| SOURCE | Mouse Monoclonal |
| CLONE | AE1/AE3 + 5D3 |
| ISOTYPE | IgG1 |
| ANTIGEN | AE1/AE3 + CK8/18 |
| LOCALIZATION | Cytoplasmic |
| POSITIVE CONTROL | Skin or adenocarcinoma |
DATASHEETS & SDS
REFERENCES
1. Seidman JD, Abbondanzo SL, Bratthauer GL. Lipid cell (steroid cell) tumor of the ovary: immunophenotype with analysis of potential pitfall due to endogenous biotinlike activity. Int J Gynecol Pathol. 1995 Oct; 14(4):331-8.
2. Bunton TE. The immunocytochemistry of cytokeratin in fish tissues. Vet Pathol. 1993 Sep; 30(5):418-425.
3. Sorensen SC, Asch BB, Connolly JL, Burstein NA, Asch HL. Structural distinctions among human breast epithelial cells revealed by the monoclonal antikeratin antibodies AEI and AE3. J Pathol. 1987 Oct; 153(2):151-162.
4. Pinkus GS, Etheridge CL, O’Connor EM. Are keratin proteins a better tumor marker than epithelial membrane antigen? A comparative immunohistochemical study of various paraffin-embedded neoplasms using monoclonal and polyclonal antibodies. Am J Clin Pathol. 1986 Mar; 85(3):269-277.
5. Pinkus GS, O’Connor EM, Etheridge CL, Corson JM. Optimal immunoreactivity of keratin proteins in formalin-fixed, paraffin-embedded tissue requires preliminary trypsinization. An immunoperoxidase study of various tumours using polyclonal and monoclonal antibodies. J Histochem Cytochem. 1985 May; 33(5):465-473.
6. Center for Disease Control Manual. Guide: Safety Management, NO. CDC-22, Atlanta, GA. April 30, 1976 “Decontamination of Laboratory Sink Drains to Remove Azide Salts.”
7. National Committee for Clinical Laboratory Standards (NCCLS). Protection of laboratory workers from infectious diseases transmitted by blood and tissue; proposed guideline. Villanova, PA 1991; 7(9). Order code M29-P.





Reviews
There are no reviews yet.