Description
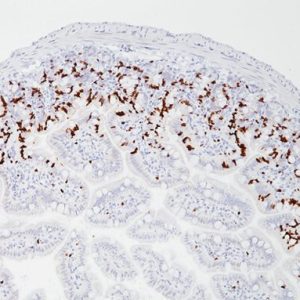
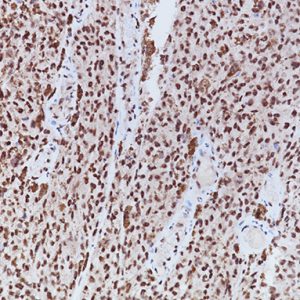
LAG-3 (Lymphocyte Activation Gene 3) is a surface receptor expressed on activated T cells, an exhaustion marker with immunosuppressive activity (1). Major histocompatibility complex class II (MHC-II) is a ligand for LAG-3; additional ligands (e.g., LSECtin and galectin-3) have also been identified (1,2). Regulatory T cells (Tregs) expressing LAG-3 have enhanced suppressive activity, whereas cytotoxic CD8+ T cells expressing LAG-3 have reduced proliferation rates and effector cytokine production in cancer and autoimmune diabetes (3,4). LAG-3+ tumor-infiltrating lymphocytes (TILs) have been reported in melanoma, colon, pancreatic, breast, lung, hematopoietic, and head and neck cancer patients (5- 11), in association with aggressive clinical features. Antibody-based LAG-3 blockade in multiple cancer mouse models restores CD8+ effector T cells and diminishes Treg populations, an effect enhanced when combined with anti-PD-1 (12). Recent studies in a metastatic ovarian cancer mouse model showed that LAG-3 blockade leads to upregulation of other immune checkpoints (PD-1, CTLA4, and TIM-3), and combination therapy targeting LAG-3, PD-1, and CTLA-4 increases functional cytotoxic T cell levels while reducing Tregs and myeloidderived suppressor cells (13,14). Multiple pre-clinical and clinical studies are testing antagonistic LAG-3 agents in combination with anti-PD-1 and/or anti-CTLA4 therapy (12-15). In view of the activating properties of soluble secreted LAG-3, a soluble agonist LAG-3 antibody (IMP321) was tested in advanced solid malignancies as a single agent (15), and demonstrated sufficient tolerability and efficacy to warrant advancement to phase II (16).
SPECIFICATIONS
Specifications
| INTENDED USE | IVD |
|---|---|
| FORMAT | Concentrate, Predilute |
| VOLUME | 0.1 ml, 0.5 ml, 6.0 ml |
| SPECIES REACTIVITY | Human |
| SOURCE | Rabbit Monoclonal |
| CLONE | CAL26 |
| ISOTYPE | IgG1 |
| ANTIGEN | Synthetic peptide derived from a region of the LAG3 protein |
| LOCALIZATION | Membrane/cytoplasm/Golgi |
DATASHEETS & SDS
REFERENCES
1. Anderson AC, et al. Lag-3, tim-3, and TIGIT: Co-inhibitory receptors with specialized functions in immune regulation. Immunity. 2016; 44:989-1004.
2. Kouo T, et al. Galectin-3 Shapes Antitumor Immune Responses by Suppressing CD8+ T Cells via LAG-3 and Inhibiting Expansion of Plasmacytoid Dendritic Cells. Cancer Immunol Res. 2015; 3:412-23.
3. Bettini M, et al. Cutting edge: accelerated autoimmune diabetes in the absence of LAG-3. J Immunol. 2011; 187:3493-8.
4. Williams JB, et al. The EGR2 targets LAG3 and 4-1BB describe and regulate dysfunctional antigen-specific CD8+ T cells in the tumor microenvironment. J Exp Med. 2017; 214:381-400.
5. Shapiro M, et al. Lymphocyte activation gene 3: a novel therapeutic target in chronic lymphocytic leukemia, Haematologica. 2017; 102:874–82.
6. Tassi E, et al. Early effector, lymphocytes coexpress multiple inhibitory receptors in primary non-small cell lung cancer. Cancer Res. 2017; 77:851–61.
7. Bottai G, et al. An immune stratification reveals a subset of PD-1/LAG-3 doublepositive triple-negative breast cancers, Breast Cancer Res. 2016; 18:121.
8. Deng WW, et al. LAG-3 confers poor prognosis and its blockade reshapes antitumor response in head and neck squamous cell carcinoma. Oncoimmunology. 2016; 5:e1239005.
9. Llosa NJ, et al. The vigorous immune microenvironment of microsatellite instable colon cancer is balanced by multiple counter-inhibitory checkpoints. Cancer Discovery. 2015; 5:43–51.
10. Meng Q, et al. Expansion of tumor-reactive T cells from patients with pancreatic cancer. J Immunother. 2016; 39:81–9.
11. Demeure CE, et al. T Lymphocytes infiltrating various tumour types express the MHC class II ligand lymphocyte activation gene-3 (LAG-3): role of LAG-3/MHC class II interactions in cell–cell contacts. Eur J Cancer. 2001; 37:1709–18.
12. Woo SR, et al. Immune inhibitory molecules LAG-3 and PD-1 synergistically regulate T-cell function to promote tumoral immune escape. Cancer Res. 2012; 72(4):917-27.
13. Huang RY, et al. LAG3 and PD1 co-inhibitory molecules collaborate to limit CD8+ T cell signaling and dampen antitumor immunity in a murine ovarian cancer model. Oncotarget. 2015; 6:27359-77.
14. Huang RY, et al. Compensatory upregulation of PD-1, LAG-3, and CTLA-4 limits the efficacy of single-agent checkpoint blockade in metastatic ovarian cancer. Oncoimmunology. 2017; 6:e1249561.
15. Baumeister SH, et al. Coinhibitory pathways in immunotherapy for cancer. Annu Rev Immunol. 2016; 34:539-73.
16. Legat A, et al. Vaccination with LAG-3Ig (IMP321) and Peptides Induces Specific CD4 and CD8 T-Cell Responses in Metastatic Melanoma Patients–Report of a Phase I/IIa Clinical Trial. Clin Cancer Res. 2016 Mar 15;22(6):1330-40.
17. Center for Disease Control Manual. Guide: Safety Management, NO. CDC-22, Atlanta, GA. April 30, 1976 “Decontamination of Laboratory Sink Drains to Remove Azide Salts.”
18. Clinical and Laboratory Standards Institute (CLSI). Protection of Laboratory Workers from Occupationally Acquired Infections; Approved Guideline-Fourth Edition CLSI document M29-A4 Wayne, PA 2014.





Reviews
There are no reviews yet.