Description
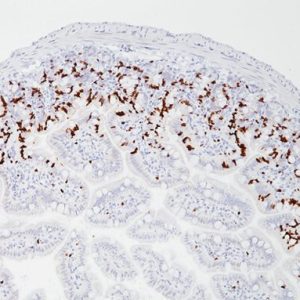
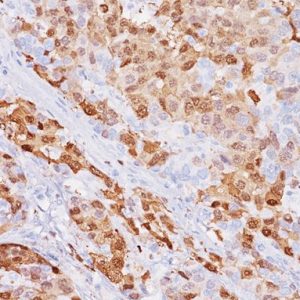
Glypican-3 (GPC3), a member of the glypican family of glycosyl phosphatidylinositol-anchored cell-surface heparin sulfate proteoglycans, plays an important role in cell growth and differentiation (1). Using the 1G12 monoclonal antibody, GPC3 has been identified as a useful tumor marker for the diagnosis of Hepatocellular Carcinoma (HCC), hepatoblastoma, melanoma, testicular germ cell tumors, and Wilms’ tumor. GPC3 protein has been shown to be expressed in most hepatocellular carcinomas (HCC), but not in normal liver nor benign hepatic lesions, including dysplastic and cirrhotic nodules (1-4). Most patients with HCC have significantly elevated serum protein levels of GPC3 (3). Several studies report that GPC3 is a sensitive diagnostic marker for HCC and a tool for differentiating HCC from non-neoplastic and pre-neoplastic liver disease (3,4). In-house TMA-based studies have shown that GPC3 is positive in 90.4% (66/73) of hepatocellular carcinoma cases, and negative in 100% of cholangiocellular carcinoma, normal liver and hyperplasia cases.
SPECIFICATIONS
Specifications
| WEIGHT | N/A |
|---|---|
| DIMENSIONS | N/A |
| INTENDED USE | IVD |
| SPECIES REACTIVITY | Human |
| SOURCE | Mouse Monoclonal |
| CLONE | 1G12 |
| ISOTYPE | IgG1 |
| ANTIGEN | Last 70 amino acids of the core protein |
| LOCALIZATION | Membrane & Cytoplasm |
| POSITIVE CONTROL | Hepatocellular Carcinoma |
DATASHEETS & SDS
INTERNATIONAL
REFERENCES
1. Kandil DH, et al., Glypican-3 A novel diagnostic marker for hepatocellular carcinoma and more. Adv Anat Pathol. 16(2): 125-9., 2009.
2. Shirakawa H, et al., Glypican-3 is a useful diagnostic marker for a component of hepatocellular carcinoma in human liver cancer. Int. J. Oncol., 34(3): 649-56, 2009.
3. Wang XY, et al., Glypican-3 expression in hepatocellular tumors: diagnostic value for preneoplastic lesions and hepatocellular carcinoma. Human Pathol., 37(11): 1435 -41. 2006.
4. Libbrecht L, et al, Glypican-3 expression distinguishes small hepatocellular carcinoma from cirrhosis, dysplastic nodules, and focal nodular hyperplasia-like nodules. Am. J. Surg. Pathol., 30(11): 1405-11. 2006.
5. Center for Disease Control Manual. Guide: Safety Management, NO. CDC-22, Atlanta, GA. April 30, 1976 “Decontamination of Laboratory Sink Drains to Remove Azide Salts.”
6. National Committee for Clinical Laboratory Standards (NCCLS). Protection of laboratory workers from infectious diseases transmitted by blood and tissue; proposed guideline. Villanova, PA 1991;7(9). Order code M29-P.





Reviews
There are no reviews yet.