Description
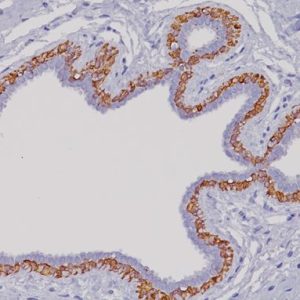
CDH17 antibody (Cadherin 17 or LI-cadherin) is a novel oncogene which is involved in tumor invasion and metastasis and is expressed in intestinal epithelium (1,2). CDH17 is a highly specific marker in colon cancer (99/99, 100%) and is a more sensitive marker than CDX2 (93/99, 94%) and CK20 (91/99, 92%) (3). Overexpression of CDH17 (and conversely, underexpression of CDX2) correlates to poor prognosis in patients with epithelial ovarian cancer (1). CDH17 may be helpful for early diagnosis of Barrett’s esophagus (4). CDH17 has been shown to be a useful marker for distinguishing between primary urinary bladder adenocarcinoma and urothelial carcinoma with glandular differentiation (5). Note that it does not distinguish primary urinary bladder adenocarcinoma from colorectal adenocarcinoma secondarily involving the bladder (5).
SPECIFICATIONS
Specifications
| WEIGHT | .4 lbs |
|---|---|
| DIMENSIONS | 1 × 1 × 1 in |
| INTENDED USE | IVD |
| SOURCE | Mouse Monoclonal |
| CLONE | 1H3 |
| ISOTYPE | IgG1/kappa |
| ANTIGEN | CDH17 |
| LOCALIZATION | Cytoplasmic and cell membrane |
| POSITIVE CONTROL | Colon Carcinoma |
DATASHEETS & SDS
REFERENCES
1. Huang LP, et al. Up-regulation of cadherin 17 and down-regulation of homeodomain protein CDX2 correlate with tumor progression and unfavorable prognosis in epithelial ovarian cancer. Int J Gynecol Cancer. 2012 Sep; 22(7):1170-6.
2. Panarelli NC, et al. Tissue-specific cadherin CDH17 is a useful marker of gastrointestinal adenocarcinomas with higher sensitivity than CDX2. Am J Clin Pathol. 2012 Aug; 138(2):211-22.
3. Tacha D, Zhou D. CDH17 is a highly specific marker and is a more sensitive marker than CDX2 and CK20 in colon cancers. Poster session presented at: CAP’14 The Pathologists’ Meeting; 2014 Sep 7-10; Chicago, IL.
4. Mokrowiecka A, et al. Liver-intestine-cadherin is a sensitive marker of intestinal differentiation during Barrett’s carcinogenesis. Dig Dis Sci. 2013 Mar; 58(3):699-705.
5. Rao Q, et al. Distinguishing primary adenocarcinoma of the urinary bladder from secondary involvement by colorectal adenocarcinoma: extended immunohistochemical profiles emphasizing novel markers. Mod Pathol. 2013 May; 26(5):725-32.
6. Center for Disease Control Manual. Guide: Safety Management, NO. CDC-22, Atlanta, GA. April 30, 1976 “Decontamination of Laboratory Sink Drains to Remove Azide Salts.”
7. Clinical and Laboratory Standards Institute (CLSI). Protection of Laboratory Workers from Occupationally Acquired Infections; Approved Guideline-Fourth Edition CLSI document M29-A4 Wayne, PA 2014.






Reviews
There are no reviews yet.